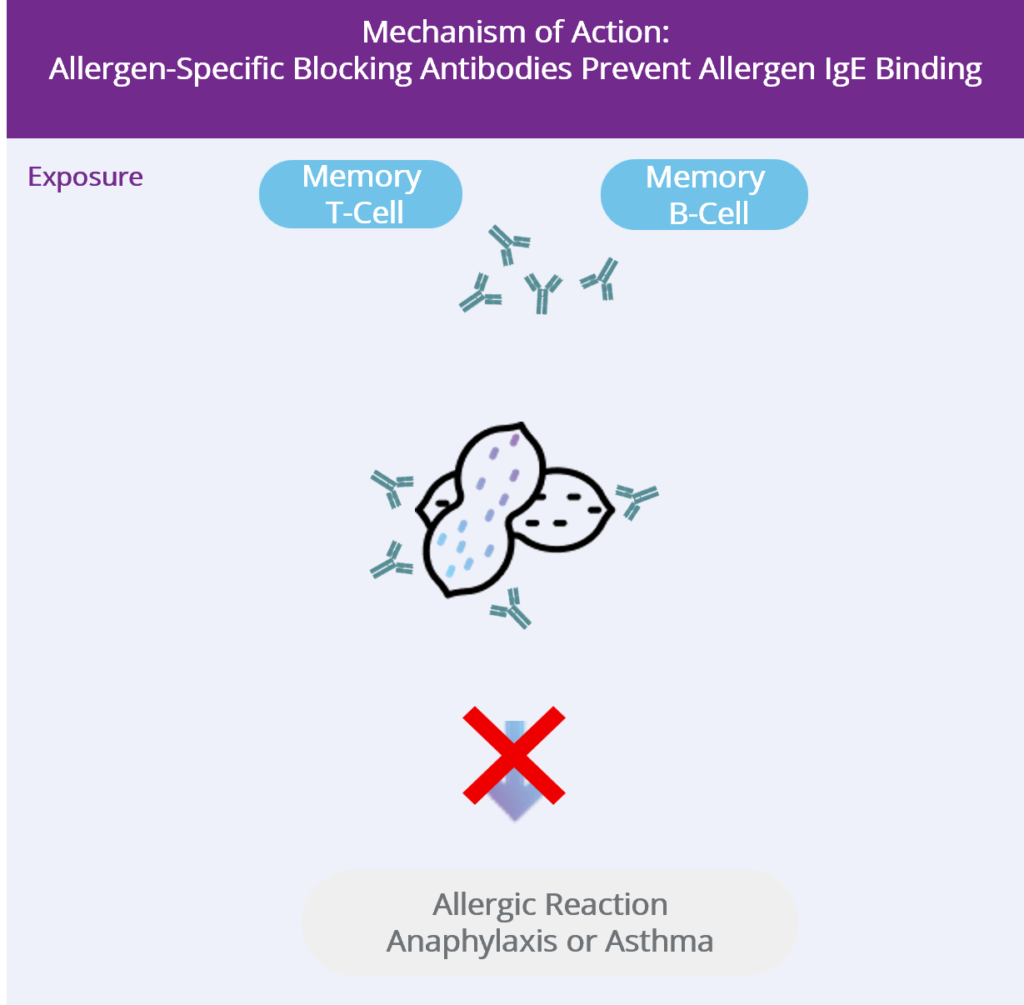
We Retrain the Immune System
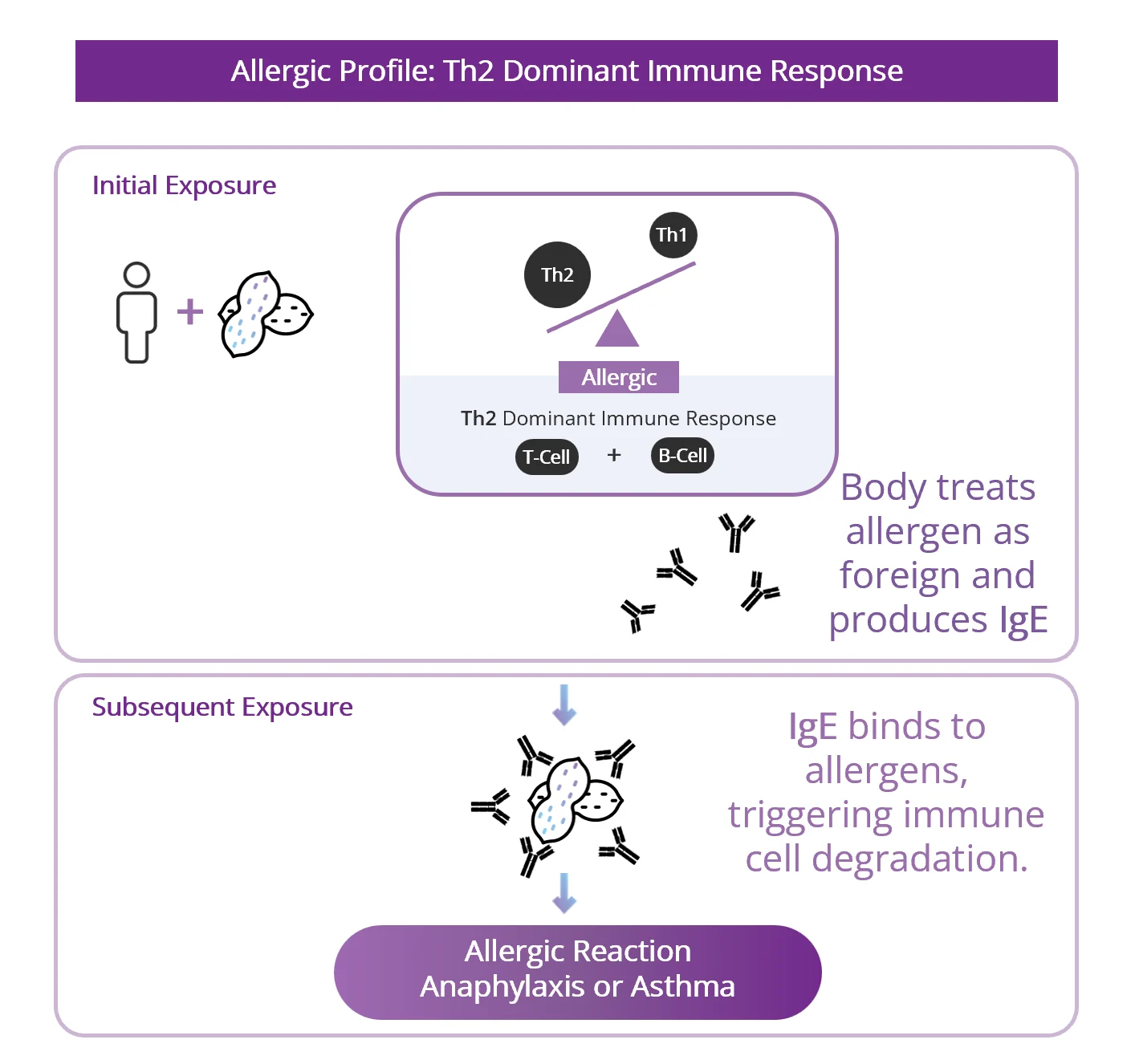
Vaccination Designed to Permanently Switch Immune Response which Drives Allergen-Specific Immune Memory
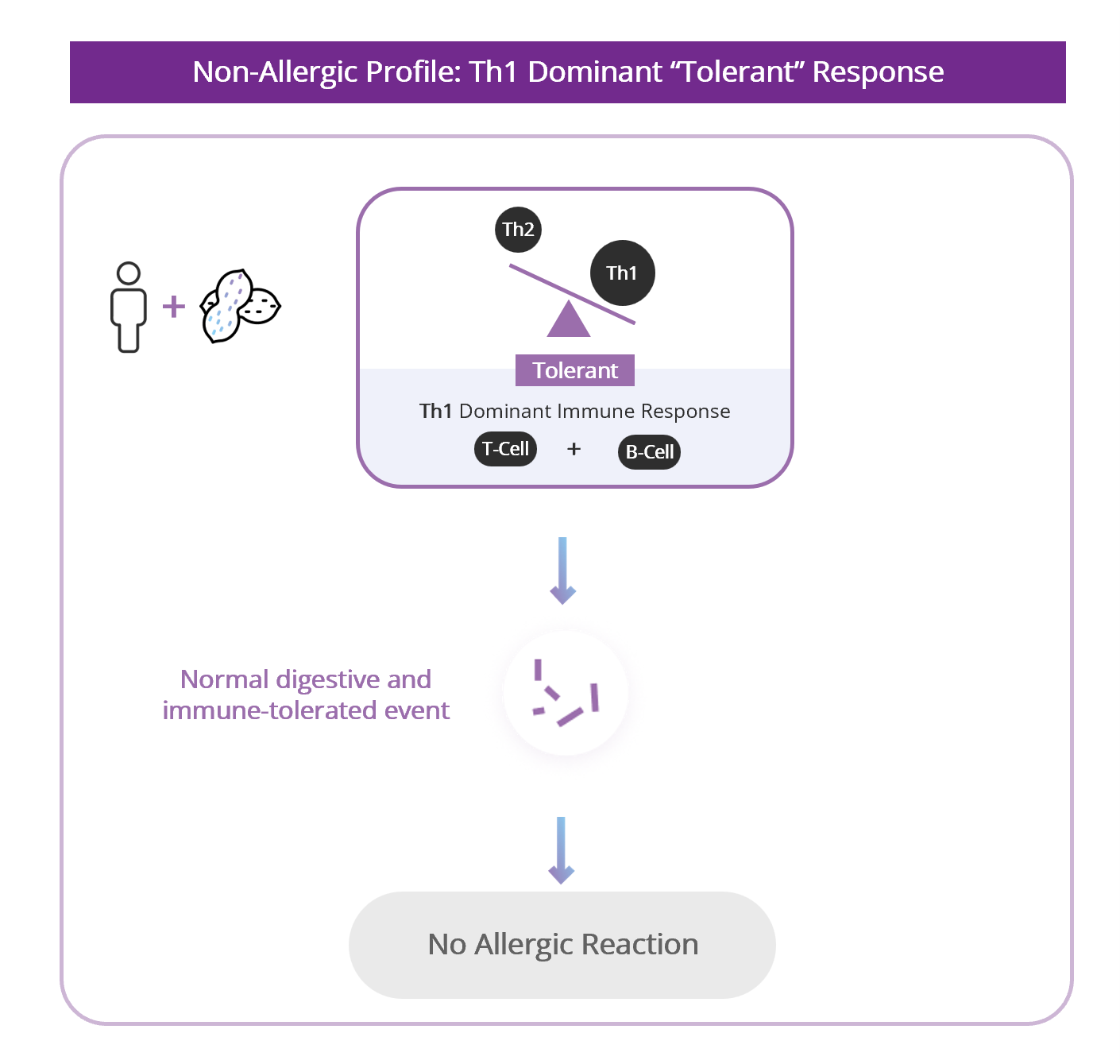
- Vaccination generates IMMUNE MEMORY
- Switch the immune response away from the allergy‑driving pathway and toward a more tolerant pathway.
- Create long‑lived immune “memory”, so the effect could last for years and potentially permanently.
- Produce “blocking antibodies” that can bind to the allergen.
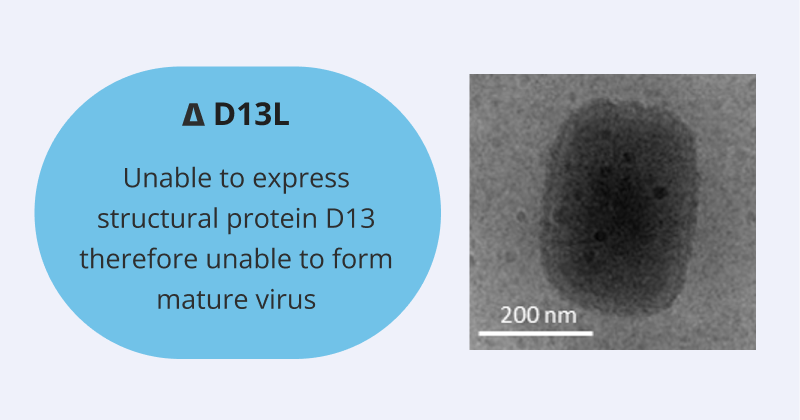
- SCV Vaccinia-Based Viral Vector Vaccine is CRITICAL to IMMUNE RETRAINING